Atoms that have the same number of protons, and hence the same atomic number, but different numbers of neutrons are called isotopes. Unlike protons, the number of neutrons is not absolutely fixed for most elements. Recall from Section 1.6 that the nuclei of most atoms contain neutrons as well as protons. German for “wolf stone” because it interfered with the smelting of tin and was thought to devour the tin doi: 10.1016/j.nuclphysa.2003.11.\) Element Symbols Based on Names No Longer in Use Elementįrom Cyprium, Latin name for the island of Cyprus, the major source of copper ore in the Roman Empire “The NUBASE Evaluation of Nuclear and Decay Properties”. Lehrbuch der Anorganischen Chemie (in German) (91–100 ed.). “Mineral Commodity Summaries 2010: Thallium” (PDF). Liddell, Henry George and Scott, Robert (eds.) “ θαλλος Archived at the Wayback Machine.”, in A Greek–English Lexicon, Oxford University Press Among the radioactive isotopes, thallium-204 is the most stable isotope and have a half-life of 3.78 years. These two isotopes make all naturally occurring thallium. Naturally occurring thallium have two stable isotopes, thalium-203 and thalium-205. Their atomic masses range from 184 to 210. 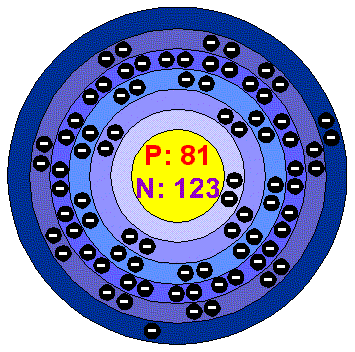
There are twenty-five isotopes of thallium. Thallium is a potential human carcinogens. These can be readily absorbed through the skin. Thallium contamination of the environment has been reported to be caused by coal-burning power plants, emission from cement factories and water contamination by leaching of thallium from ore processing plants. Thallium compounds are highly soluble in water that increases their risk of toxicity. However, due to the dangers of their non-selective poisoning, the use of thallium-based poisons has been banned in various countries. They have been used as insecticides and rat poison. The radioactive isotope thallium-201 is used for nuclear medicine scan.Ĭertain compounds of thallium are highly toxic.Thallium is used as detectors in infrared devices.Thallium is used in pharmaceutical industry to make various drugs and medicines.It is used as photoresistors, rectifiers and in gamma radiation detection devices. Thallium is widely used in making various electronics, and about 70% of the thallium produced world wide is used in this industry.It dissolves in nitric acid and sulfuric acid and forms salts of nitrate and sulphate. thallium reacts with water to form thallium hydroxide. Thallium reacts with halogens and thallium halides are stable in nature. Thallium compounds with +3 oxidation state are strong oxidizing agents. Thallium exists in two oxidation states, +3 and +1. Thallium has a high boiling point, 1473 ° C and a melting point of 304 ☌. It is quite dense and have a density of 11.85 g/cm 3. Freshly isolated thallium looks like tin and is readily discolored (becomes bluish-grey) and loses it shine when exposed to air. Thallium is a greyish color post-transition metal. Large deposits of thallium are found in southern Macedonia where active mining of thallium is carried out. Annually, about 10 metric tons of thallium is produced worldwide as a byproduct during the smelting of lead, zinc and copper ores. It is also found in ocean beds in combination with manganese nodules. The mineral crookesite, lorandite (TlAsS 2) and hutchinsonite (TlPbAs 5S 9) are common minerals of thallium. Commercially, thallium is extracted from its sulfide-ores of heavy metals, such as zinc, lead, copper etc., which contain 16% to 60 of thallium. it mostly exists in the form of ores with other elements, mostly potassium. Thallium does not exist in free elemental form in nature. Thallium is an abundant element and is present in about 0.7 mg/kg of the Earth’s crust. The name thallium was given by Crookes which has been derived from the Greek word thallos which means green twig or shoot. Thallium gave distinct green spectral lines in the spectroscopy analysis. They used flame spectroscopy for the discovery of the novel element. Thallium was discovered by two chemists, Claude-Auguste Lamy and William Crookes, in 1986 who were independently working with sulfuric acid. Due to its toxic nature, it has been historically used as ant killer and rat poison. Thallium was discovered independently in 1986 by William Crookes and Claude-Auguste Lamy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
